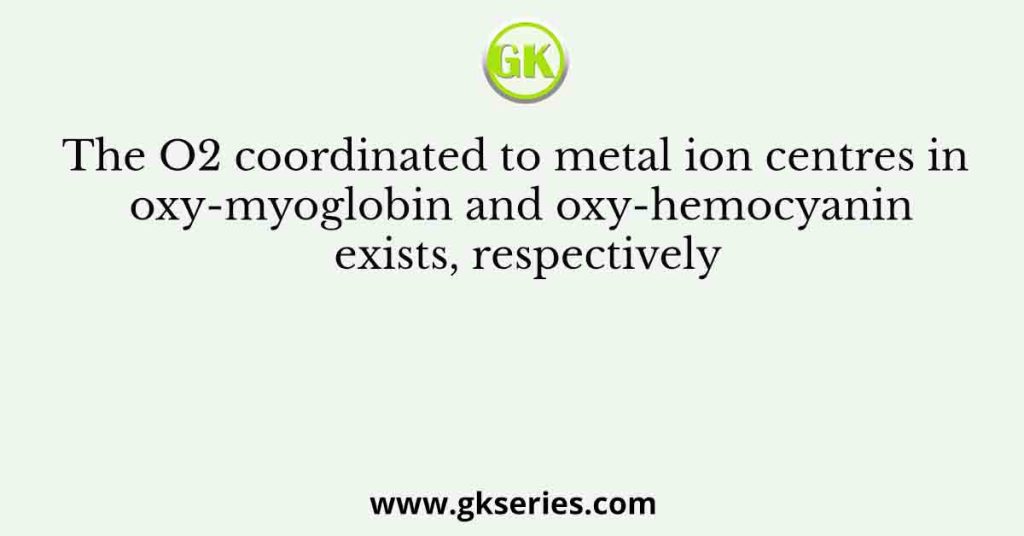The reaction of equimolar quantities of Fe(CO)5 and OH– gives a complex species X which on further reaction with MnO2
Q. The reaction of equimolar quantities of Fe(CO)5 and OH– gives a complex species X which on further reaction with MnO2 gives species Y. X and Y, respectively, are (A) [Fe(CO)5(OH)]– and Fe2(CO)9 (B) [Fe(CO)4]2– and Mn2(CO)10 (C) [HFe(CO)4]– and Fe2O3 (D) [HFe(CO)4]– and Fe3(CO)12 Ans: [HFe(CO)4]- and Fe3(CO)12



![Second-order rate constant for the reaction between [Co(NH3)5X]n+ (n = 3 for X = NH3 and H2O](https://www.gkseries.com/blog/wp-content/uploads/2023/09/Second-order-rate-constant-for-the-reaction-between-CoNH35Xn-n-3-for-X-NH3-and-H2O-1024x536.jpg)

![Spectroscopic ground state term symbols of cobalt ions in [Co(H2O)6]2+ and [CoCl4]2–](https://www.gkseries.com/blog/wp-content/uploads/2023/09/Spectroscopic-ground-state-term-symbols-of-cobalt-ions-in-CoH2O62-and-CoCl42–-1024x536.jpg)