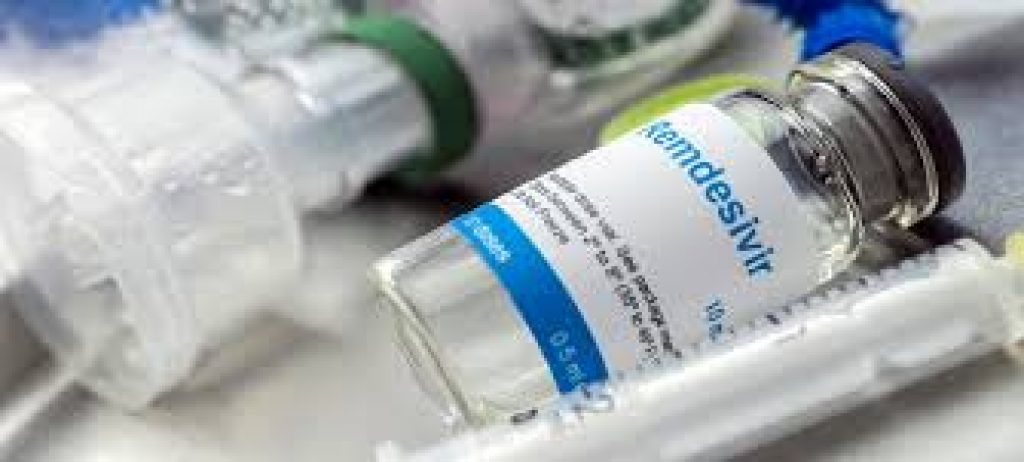
US FDA allowed use of remdesivir drug for severely ill COVID-19 patients
U.S. Food and Drug Administration issued an emergency use authorization for the investigational antiviral drug remdesivir for the treatment of suspected or laboratory-confirmed COVID-19 in adults and children hospitalized with severe disease. A study by Gilead Sciences in the US showed that Remdesivir shortens the recovery time by 31% or about 4 days on average, […]
US FDA allowed use of remdesivir drug for severely ill COVID-19 patients Read More »