Questions
1
Hydrogen resembles halogens in many respects for which several factors are responsible. Of the following factors which one is most important in this respect?
Answer:Its tendency to gain a single electron in its valence shell to attain stable electronic configuration.
2
Why does H+ ion always get associated with other atoms or molecules?
Answer:Loss of an electron from hydrogen atom results in a nucleus of very small size as compared to other atoms or ions. Due to smal size it cannot exist free.
3
Metal hydrides are ionic, covalent or molecular in nature. Among LiH, NaH, KH, RbH, CsH, the correct order of increasing ionic character is
Answer:LiH < NaH < KH < RbH < CsH
5
Radioactive elements emit α, β and γ rays and are characterised by their halflives. The radioactive isotope of hydrogen is
Answer:Tritium
6
Consider the reactions
(A) H2O2 + 2HI → I2 + 2H2O (B) HOCl + H2O2 → H3O+ + Cl+ O2
Which of the following statements is correct about H2O2 with reference to these reactions? Hydrogen perioxide is ________.
Answer:an oxidising agent in (A) and reducing agent in (B)
7
The oxide that gives H2O2 on treatment with dilute H2SO4 is —
Answer:BaO2 .8H2O + O2
8
Which of the following reactions increases production of dihydrogen from synthesis gas?
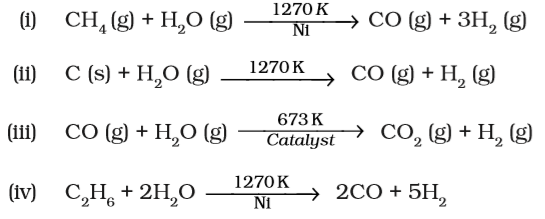
Answer:(iii)
9
When sodium peroxide is treated with dilute sulphuric acid, we get ______.
Answer:sodium sulphate and hydrogen peroxide
10
Hydrogen peroxide is obtained by the electrolysis of ______.
Answer:sulphuric acid
11
Which of the following reactions is an example of use of water gas in the synthesis of other compounds?
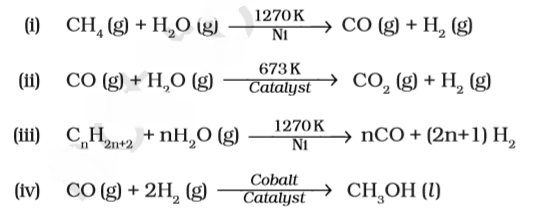
Answer:(iv)
12
Which of the following ions will cause hardness in water sample?
Answer:Ca2+
13
Which of the following compounds is used for water softening?
Answer:Na6P6O18
14
Elements of which of the following group(s) of periodic table do not form hydrides.
Answer:Groups 7, 8, 9
17
Dihydrogen can be prepared on commercial scale by different methods. In its preparation by the action of steam on hydrocarbons, a mixture of CO and H2 gas is formed. It is known as
Answer:A, B
18
Which of the following statement(s) is/are correct in the case of heavy water?
Answer:A, C
20
Some of the properties of water are described below. Which of them is/are not correct?
Answer:C, D
