Questions
1
The element which exists in liquid state for a wide range of temperature and can be used for measuring high temperature is
Answer:Ga
3
The geometry of a complex species can be understood from the knowledge of type of hybridisation of orbitals of central atom. The hybridisation of orbitals of central atom in [Be(OH)4]– and the geometry of the complex are respectively
Answer:sp3 , tetrahedral
5
The exhibition of highest co-ordination number depends on the availability of vacant orbitals in the central atom. Which of the following elements is not likely to act as central atom in 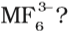
Answer:B
6
Boric acid is an acid because its molecule
Answer:accepts OH– from water releasing proton
7
Catenation i.e., linking of similar atoms depends on size and electronic configuration of atoms. The tendency of catenation in Group 14 elements follows the order:
Answer:C >> Si > Ge ≈ Sn
8
Silicon has a strong tendency to form polymers like silicones. The chain length of silicone polymer can be controlled by adding
Answer:Me3SiCl
9
onisation enthalpy (∆i H1 kJ mol–1) for the elements of Group 13 follows the order.
Answer:B > Al < Ga > In < Tl
10
In the structure of diborane
Answer:2 boron atoms and 4 terminal hydrogen atoms lie in the same plane and 2 bridging hydrogen atoms lie in the perpendicular plane.
11
A compound X, of boron reacts with NH3 on heating to give another compound Y which is called inorganic benzene. The compound X can be prepared by treating BF3 with Lithium aluminium hydride. The compounds X and Y are represented by the formulas.
Answer:B2H6 , B3N3H6
12
Quartz is extensively used as a piezoelectric material, it contains ___________.
Answer:Si
15
Cement, the important building material is a mixture of oxides of several elements. Besides calcium, iron and sulphur, oxides of elements of which of the group (s) are present in the mixture?
Answer:groups 2, 13 and 14
18
Me3SiCl is used during polymerisation of organo silicones because
Answer: A, B
20
Which of the following statements are correct. Answer on the basis of Fig
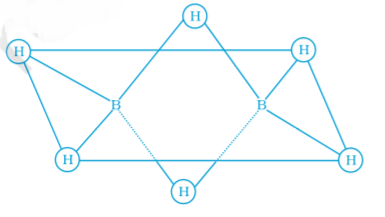
Answer:A, B, D
