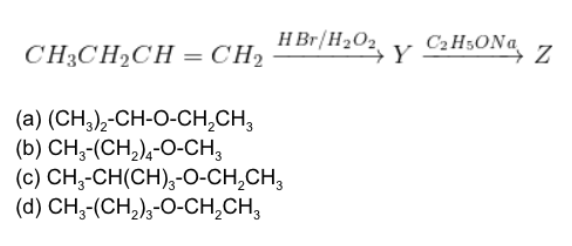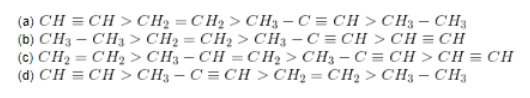Questions
1
Assertion (A) : Toluene on Friedal Crafts methylation gives o– and p–xylene.
Reason (R) : CH3-group bonded to benzene ring increases electron density at o– and p– position.
Answer:Both A and R are correct and R is the correct explanation of A.
2
Assertion (A) : Nitration of benzene with nitric acid requires the use of concentrated sulphuric acid.
Reason (R) : The mixture of concentrated sulphuric acid and concentrated nitric acid produces the electrophile, NO2+ .
Answer:Both A and R are correct and R is the correct explanation of A.
3
Assertion (A) : Among isomeric pentanes, 2, 2-dimethylpentane has highest boiling point.
Reason (R) : Branching does not affect the boiling point.
Answer:Both A and R are not correct.
4
Arrange the following hydrogen halides in order of their decreasing reactivity with propane.
Answer:HI > HBr > HCl
5
The increasing order of reduction of alkyl halides with zinc and dilute HCl is
Answer:R–Cl < R–Br < R–I
6
Arrange the halogens F2 , Cl2 , Br2 , I2 , in order of their increasing reactivity with alkanes.
Answer:I2 < Br2 < Cl2 < F2
7
Arrange the following in decreasing order of their boiling points.
(A) n–butane (B) 2–methylbutane
(C) n-pentane (D) 2,2–dimethylpropane
Answer:C > B > D > A
9
C6H5CHO is formed when C6H6 is treated with CO and HCl in the presence of anhydrous AlCl3. Name of the reaction is:
Answer:Gattermann Koch reaction
10
Isopropyl iodide is formed after reaction of propene with HI, this is due to
Answer:more stable carbocation
11
Which among the following reagent can be used to distinguish between CH2BrCH=CH2 from CH3CH=CHBr?
Answer:AgNO3/C2H5OH
12
Which one of the following compound forms salt on reaction with NaNH2?
Answer:C2H2
13
Which one of the following halide can be used in the Friedel-Crafts reaction?
Answer:Isopropyl chloride
14
Choose the process by which liquid hydrocarbons can be converted to gaseous hydrocarbons
Answer:cracking
15
The product obtained on heating 2-Bromopentane with potassium ethoxide in ethanol is
Answer:trans-2-pentene
18
Among the following compounds the one that is most reactive towards electrophilic nitration is
Answer:Toluene
19
Which branched chain isomer of the hydrocarbon with molecular mass 72u gives only one isomer of mono substituted alkyl halide?
Answer:Neopentane